Introduction
Radiofrequency catheter ablation (RFCA) is among the most effective treatments for atrial fibrillation (AF) [1–3]. Clinical trials have provided comprehensive evidence supporting the safety and effectiveness of RFCA [4–8]. However, studies on patients receiving RFCA have reported AF recurrence rates ranging from 23% to 43% [9]. The cornerstone of RFCA is circumferential pulmonary vein isolation (CPVI) [10]. Composite ablation based on CPVI (com-CPVI), which includes linear ablation [11], substrate modification [12], and vein of Marshall ethanol infusion [13], may enhance the success rate of RFCA. However, in general, AF recurrence after RFCA cannot be overlooked.
According to the most recent consensus among Chinese AF experts, the “blanking period” is defined as the 3-month period after RFCA. Approximately 60% of AF recurrence during this period is believed to resolve spontaneously and should not be considered clinically significant [3]. However, previous studies have demonstrated that AF recurrence during the blanking period independently predicts long-term AF recurrence after RFCA [14, 15]. Immediate recurrence (Im-Recurr) is defined herein as AF recurrence within 48 hours following ablation. Because Im-Recurr represents a specific subset of AF recurrence during the blanking period, we hypothesized that it might have value in predicting long-term AF recurrence. Moreover, we reasoned that Im-Recurr during hospitalization might potentially lead to increased costs and extended hospital stays.
Currently, no research available on the medical economic effects, predictors, and outcomes of Im-Recurr. Consequently, this study was aimed at investigating these issues by conducting a retrospective analysis at a single center.
Methods
The data supporting the findings of this study are available from the corresponding author on reasonable request.
Ethical Statement
This study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (approval number: 2022-918-Fast), and was registered in the Chinese Clinical Trial Registry under registration number ChiCTR2200065235. Because of this study’s retrospective and observational nature, patients’ personal information was not revealed; therefore, our institutional ethics committee approved exemption from informed consent requirements.
Study Population
Inclusion criteria: This study included consecutive patients with AF who underwent RFCA between July 1, 2021, and June 30, 2022.
Exclusion criteria: This study excluded patients who did not undergo CPVI during RFCA (usually patients who experienced AF recurrence and received a second RFCA), and patients with unsuccessful sinus conversion during RFCA or intraoperative electric cardioversion (IEC). Regardless of their underlying disease (such as cancer or renal insufficiency), patients who were assessed by a physician, deemed suitable, and received RFCA treatment were included in the analysis.
Data Acquisition
Patients’ information was retrieved from our center’s electronic medical record system. In the analysis, the patients were grouped according to the following variables and definitions.
①Demographic characteristics and medical history: sex, age, body mass index, hypertension, diabetes, heart failure, stroke, vascular disease, cancer, cardiomyopathy, and abnormal uric acid metabolism [16].
②Laboratory and imaging findings: transesophageal echocardiography findings of spontaneous echocardiographic contrast; transthoracic echocardiography measurements of the left atrial anteroposterior diameter and left ventricular ejection fraction; and laboratory parameters including hemoglobin, serum uric acid, creatinine, cholesterol, albumin, homocysteine, and C-reactive protein levels.
③RFCA related information and perioperative medication: AF type (paroxysmal or persistent), determined according to the medical history and diagnosis; RFCA strategies, determined according to operation records (group 1: CPVI; group 2: com-CPVI). Com-CPVI was defined as one or more additional ablation methods (linear ablation, substrate modification, and vein of Marshall ethanol infusion) based on CPVI. Whether patients received IEC was recorded, given that some patients may show AF rhythm after successful CPVI or com-CPVI, and later convert to sinus rhythm through IEC. Perioperative medications included amiodarone, dronedarone, propafenone, beta blockers, sacubitril valsartan, sodium-dependent glucose transporter 2 inhibitor, angiotensin converting enzyme inhibitor/angiotensin receptor blocker, and statins.
④Endpoints: recurrence of AF, defined as AF, atrial flutter, or atrial tachycardia with a duration of 30 seconds or more [3]. The presence of Im-Recurr was confirmed within 48 hours after RFCA according to medical and nursing records, postoperative cardiac monitoring, and 24-hour Holter examination. In addition, costs, hospitalization durations, and hospitalization durations after ablation were recorded. For Im-Recurr, physicians made a comprehensive judgment based on patients’ symptoms and other conditions, then decided whether to perform electric cardioversion, medical cardioversion, or no special treatment.
Follow Up
AF recurrence 3 months after RFCA rather than in the blanking period, is considered to indicate true recurrence [3]. On November 30, 2022, we retrieved the follow-up and reexamination records for all patients from our center’s electronic medical record system. Patients with a follow-up time not exceeding 90 days after RFCA were considered lost to follow-up and were excluded from the analysis. We evaluated whether patients experienced AF recurrence according to their medical records, electrocardiograms, and/or 24-hour Holter monitoring results. The longest follow-up time was recorded if no AF recurrence occurred; otherwise, the earliest occurrence of AF recurrence after 90 days of RFCA was recorded.
Statistical Analysis
The expectation maximization algorithm was used to evaluate missing data (Supplementary Table 1). Continuous variables are represented as means ± standard deviations, and differences between groups were compared with Student’s t-test. Categorical variables are described as frequencies (and percentages), and differences were compared with the chi-square test (SPSS 26.0).
Regression analysis: Multiple linear regression analysis was performed to analyze the factors influencing medical economic indicators, and binary logistic regression analysis was used to analyze the factors influencing Im-Recurr. Preliminary regression analyses were separately conducted on the following datasets: (1) demographic characteristics and medical history, (2) laboratory and imaging findings, and (3) RFCA related information and perioperative medication. Variables with a P-value <0.1 in the preliminary analyses were selected for formal regression analysis. B represents the regression coefficient, SE represents the standard error, OR represents the odds ratio, and CI represents the confidence interval (SPSS 26.0).
Propensity score matching (PSM) analysis: 1:1 PSM without replacement was used to adjust for differences. A caliper width equal to 0.2 of the standard deviation of the logit of the propensity score was used. The variables associated with demographic characteristics and medical history were used for matching. For PSM analysis, statistical tools from the Faculty of Science, Department of Statistics, University of British Columbia (https://www.stat.ubc.ca/~rollin/stats/ssize), were used to evaluate the power of the sample size, with a one-sided α of 0.05 (STATA 17.0).
Survival curves were plotted, and differences between groups were evaluated with the log-rank (Mantel-Cox) test (GraphPad Prism 8.0.2).
Statistical significance is indicated as follows: * for P < 0.05, ** for P < 0.01, and *** for P < 0.001.
Results
Characteristics of the Study Population
A total of 904 patients met the inclusion criteria. Among them, five patients who did not receive CPVI were excluded. Those five patients experienced AF recurrence after previous RFCA and underwent RFCA only for the trigger site. One patient continued to show AF rhythm after RFCA and IEC, and ultimately opted against sinus conversion and consequently was excluded from the analysis. Therefore, the final analysis included 898 patients.
Among the 898 included patients, 128 (14.3%) experienced Im-Recurr after RFCA (Table 1). In this Im-Recurr group, the prevalence of persistent AF was 47.7%, a percentage significantly higher than the 36.8% in the control group (P = 0.019). Additionally, 50.8% of the Im-Recurr group received IEC during RFCA, as compared with 37.9% of the control group (P = 0.006). Furthermore, significant statistical differences were observed in sex, age, and left atrial anteroposterior diameter between groups (Table 1).
Comparison of Baseline Characteristics between the Im-Recurr and Control Groups.
| Total N = 898 | Im-Recurr N = 128 | Control N = 770 | P value | |
|---|---|---|---|---|
| Demographic characteristics and medical history | ||||
| Male | 585 (65.1) | 72 (56.3) | 513 (66.6) | 0.023* |
| Age, years | 62.1 ± 10.8 | 64.0 ± 9.4 | 61.8 ± 11.0 | 0.020* |
| Body mass index, kg/m2 | 24.3 ± 3.8 | 24.5 ± 3.6 | 24.2 ± 3.8 | 0.412 |
| Hypertension | 481 (53.6) | 75 (58.6) | 406 (52.7) | 0.218 |
| Diabetes | 112 (12.5) | 19 (14.8) | 93 (12.1) | 0.380 |
| Heart failure | 116 (12.9) | 18 (14.1) | 98 (12.7) | 0.677 |
| Stroke | 63 (7.0) | 13 (10.2) | 50 (6.5) | 0.133 |
| Vascular disease | 161 (17.9) | 23 (18.0) | 138 (17.9) | 0.990 |
| Cancer | 43 (4.8) | 5 (3.9) | 38 (4.9) | 0.614 |
| Cardiomyopathy | 32 (3.6) | 7 (5.5) | 25 (3.2) | 0.318 |
| Abnormal uric acid metabolism | 222 (24.7) | 37 (28.9) | 185 (24.0) | 0.236 |
| Laboratory and imaging findings | ||||
| Spontaneous echocardiographic contrast | 95 (10.7) | 16 (12.6) | 79 (10.4) | 0.463 |
| LAD, cm | 4.0 ± 0.7 | 4.1 ± 0.6 | 4.0 ± 0.7 | 0.043* |
| Ejection fraction, % | 63.3 ± 8.6 | 62.8 ± 8.8 | 63.4 ± 8.6 | 0.472 |
| Hemoglobin, g/L | 141.4 ± 16.7 | 140.9 ± 17.4 | 141.5 ± 16.6 | 0.690 |
| Uric acid, μmol/L | 349.0 ± 90.8 | 347.5 ± 87.7 | 349.3 ± 91.4 | 0.837 |
| Creatinine, μmol/L | 82.7 ± 39.7 | 78.4 ± 17.5 | 83.4 ± 42.2 | 0.192 |
| Cholesterol, mmol/L | 4.1 ± 1.0 | 4.1 ± 1.0 | 4.1 ± 1.0 | 0.869 |
| Albumin, g/L | 41.5 ± 3.7 | 41.2 ± 3.7 | 41.6 ± 3.7 | 0.297 |
| Homocysteine, μmol/L | 11.9 ± 5.3 | 12.1 ± 5.4 | 11.9 ± 5.3 | 0.690 |
| C-rective protein, mg/L | 2.7 ± 8.3 | 4.0 ± 16.6 | 2.4 ± 5.9 | 0.303 |
| RFCA related information and perioperative medication | ||||
| Persistent AF | 344 (38.3) | 61 (47.7) | 283 (36.8) | 0.019* |
| Com-CPVI | 298 (33.2) | 39 (30.5) | 259 (33.6) | 0.481 |
| IEC | 357 (39.8) | 65 (50.8) | 292 (37.9) | 0.006** |
| Amiodarone | 797 (88.8) | 112 (87.5) | 685 (89.0) | 0.628 |
| Dronedarone | 49 (5.5) | 6 (4.7) | 43 (5.6) | 0.679 |
| Propafenone | 29 (3.2) | 5 (3.9) | 24 (3.1) | 0.843 |
| Beta blockers | 269 (30.0) | 45 (35.2) | 224 (29.1) | 0.165 |
| Sacubitril valsartan | 101 (11.2) | 14 (10.9) | 87 (11.3) | 0.905 |
| SGLT2I | 13 (1.4) | 1 (0.8) | 12 (1.6) | 0.778 |
| ACEI/ARB | 205 (22.8) | 36 (28.1) | 169 (21.9) | 0.123 |
| Statins | 315 (35.1) | 45 (35.2) | 270 (35.1) | 0.984 |
| Medical economic indicators | ||||
| Cost, 103 CNY | 77.4 ± 8.7 | 81.0 ± 16.4 | 76.8 ± 6.4 | 0.004** |
| Duration of hospitalization, days | 5.2 ± 2.1 | 6.1 ± 2.2 | 5.0 ± 2.1 | <0.001*** |
| Duration of hospitalization after ablation, days | 2.9 ± 1.4 | 3.8 ± 1.9 | 2.8 ± 1.3 | <0.001*** |
LAD: Left atrial anteroposterior diameter; SGLT2I: Sodium-dependent glucose transporter 2 inhibitor; ACEI: Angiotensin converting enzyme inhibitor; ARB: Angiotensin receptor blocker.
*P < 0.05, **P < 0.01 and ***P < 0.001.
Medical Economic Analysis of Im-Recurr
Analysis of the baseline characteristics (Table 1) indicated that the Im-Recurr group had higher hospitalization costs (81.0 ± 16.4 vs. 76.8 ± 6.4, P = 0.004, unit: CNY), longer hospitalization durations (6.1 ± 2.2 vs. 5.0 ± 2.1, P < 0.001, unit: days), and longer hospitalization durations after ablation (3.8 ± 1.9 vs. 2.8 ± 1.3, P < 0.001, unit: days) than the control group.
Multiple linear regression analysis was conducted to explore the factors influencing the medical economic indicators (Table 2 and Supplementary Tables 2–4). Im-Recurr significantly increased costs (B = 3.893, 95% CI: [2.298, 5.487], P < 0.001), hospitalization durations (B = 0.933, 95% CI: [0.549, 1.317], P < 0.001), and hospitalization durations after ablation (B = 0.968, 95% CI: [0.709, 1.228], P < 0.001). Age, cardiomyopathy and heart failure status, levels of C-reactive protein and albumin, and statin use also affected the costs and hospitalization durations (Table 2).
Multiple Linear Regression Analysis of Medical Economic Indicators.
| B | SE | 95% CI | P value | |
|---|---|---|---|---|
| Cost | ||||
| Age | 0.041 | 0.027 | (−0.012, 0.094) | 0.132 |
| Heart failure | 1.093 | 0.898 | (−0.670, 2.856) | 0.224 |
| Cardiomyopathy | 5.752 | 1.575 | (2.660, 8.844) | <0.001*** |
| Spontaneous echocardiographic contrast | 1.757 | 0.938 | (−0.084, 3.598) | 0.061 |
| C-rective protein | 0.110 | 0.035 | (0.042, 0.178) | 0.002** |
| Statins | 1.274 | 0.621 | (0.056, 2.493) | 0.040* |
| Im-Recurr | 3.893 | 0.812 | (2.298, 5.487) | <0.001*** |
| Duration of hospitalization | ||||
| Age | 0.007 | 0.007 | (−0.006, 0.020) | 0.295 |
| Heart failure | 0.461 | 0.213 | (0.043, 0.879) | 0.031* |
| Stroke | 0.356 | 0.272 | (−0.179, 0.890) | 0.192 |
| Vascular disease | 0.319 | 0.188 | (−0.050, 0.689) | 0.090 |
| Cardiomyopathy | 0.906 | 0.377 | (0.166, 1.645) | 0.016* |
| Albumin | −0.062 | 0.019 | (−0.099, −0.024) | 0.001** |
| C-rective protein | 0.009 | 0.008 | (−0.007, 0.025) | 0.282 |
| Persistent AF | 0.236 | 0.143 | (−0.044, 0.517) | 0.099 |
| Statins | 0.554 | 0.158 | (0.244, 0.863) | <0.001*** |
| Im-Recurr | 0.933 | 0.196 | (0.549, 1.317) | <0.001*** |
| Duration of hospitalization after ablation | ||||
| Age | 0.011 | 0.005 | (0.002, 0.020) | 0.018* |
| Heart failure | 0.279 | 0.143 | (−0.002, 0.560) | 0.052 |
| Spontaneous echocardiographic contrast | 0.291 | 0.152 | (−0.007, 0.589) | 0.056 |
| Albumin | −0.028 | 0.013 | (−0.053, −0.003) | 0.027* |
| Statins | 0.140 | 0.100 | (−0.055, 0.336) | 0.159 |
| Im-Recurr | 0.968 | 0.132 | (0.709, 1.228) | <0.001*** |
*P < 0.05, **P < 0.01 and ***P < 0.001.
The above results indicated that Im-Recurr was associated with elevated costs and hospitalization durations among patients with AF who underwent RFCA.
Logistic Regression Analysis of Predictors of Im-Recurr
Binary logistic regression analysis indicated that age (OR = 1.034, 95% CI: [1.008, 1.060], P = 0.010), RFCA strategies (OR = 0.488, 95% CI: [0.284, 0.839], P = 0.009), and IEC (OR = 2.120, 95% CI: [1.263, 3.561], P = 0.004) were significantly associated with the risk of Im-Recurr (Table 3 and Supplementary Table 5).
Logistic Regression Analysis of Im-Recurr.
| B | SE | OR | 95% CI | P value | |
|---|---|---|---|---|---|
|
| |||||
| Im-Recurr | |||||
| Male | −0.308 | 0.264 | 0.735 | (0.438, 1.233) | 0.243 |
| Age | 0.033 | 0.013 | 1.034 | (1.008, 1.060) | 0.010* |
| Cardiomyopathy | 0.779 | 0.549 | 2.179 | (0.742, 6.395) | 0.156 |
| Spontaneous echocardiographic contrast | 0.167 | 0.350 | 1.182 | (0.595, 2.350) | 0.633 |
| LAD | 0.061 | 0.200 | 1.063 | (0.718, 1.574) | 0.761 |
| Creatinine | −0.007 | 0.007 | 0.993 | (0.980, 1.006) | 0.298 |
| Homocysteine | 0.013 | 0.023 | 1.013 | (0.968, 1.060) | 0.571 |
| Com-CPVI | −0.716 | 0.276 | 0.488 | (0.284, 0.839) | 0.009** |
| IEC | 0.752 | 0.264 | 2.120 | (1.263, 3.561) | 0.004** |
| Amiodarone | −0.449 | 0.416 | 0.638 | (0.282, 1.444) | 0.281 |
| Dronedarone | −0.713 | 0.659 | 0.490 | (0.135, 1.783) | 0.279 |
LAD: Left atrial anteroposterior diameter.
*P < 0.05 and **P < 0.01.
Psm Analysis of Predictors of Im-Recurr
Logistic regression analysis indicated that IEC was associated with Im-Recurr risk. To further explore this correlation, we conducted PSM analysis to compare the incidence of Im-Recurr between patients who underwent IEC and those who did not undergo IEC during RFCA (Table 4).
Comparison of Patients with or Without IEC by PSM Analysis.
| Before PSM | After PSM | |||||
|---|---|---|---|---|---|---|
| With IEC N = 357 | Without IEC N = 541 | P value | With IEC N = 313 | Without IEC N = 313 | P value | |
| Demographic characteristics and medical history | ||||||
| Male | 244 (68.3) | 341 (63.0) | 0.102 | 212 (67.7) | 214 (68.4) | 0.864 |
| Age, years | 63.0 ± 9.8 | 61.6 ± 11.5 | 0.057 | 62.7 ± 9.7 | 62.1 ± 10.9 | 0.527 |
| Body mass index, kg/m2 | 24.8 ± 3.7 | 23.9 ± 3.8 | <0.001*** | 24.7 ± 3.0 | 24.8 ± 3.3 | 0.599 |
| Hypertension | 200 (56.0) | 281 (51.9) | 0.230 | 172 (55.0) | 170 (54.3) | 0.872 |
| Diabetes | 37 (10.4) | 75 (13.9) | 0.120 | 37 (11.8) | 38 (12.1) | 0.902 |
| Heart failure | 69 (19.3) | 47 (8.7) | <0.001*** | 35 (11.2) | 44 (14.1) | 0.279 |
| Stroke | 21 (5.9) | 42 (7.8) | 0.280 | 20 (6.4) | 17 (5.4) | 0.611 |
| Vascular disease | 68 (19.0) | 93 (17.2) | 0.478 | 59 (18.8) | 50 (16.0) | 0.343 |
| Cancer | 10 (2.8) | 33 (6.1) | 0.023* | 10 (3.2) | 8 (2.6) | 0.632 |
| Cardiomyopathy | 14 (3.9) | 18 (3.3) | 0.638 | 8 (2.6) | 12 (3.8) | 0.363 |
| Abnormal uric acid metabolism | 107 (30.0) | 115 (21.3) | 0.003** | 85 (27.2) | 89 (28.4) | 0.721 |
| Laboratory and imaging findings | ||||||
| Spontaneous echocardiographic contrast | 66 (18.6) | 29 (5.5) | <0.001*** | 59 (19.0) | 22 (7.1) | <0.001*** |
| LAD, cm | 4.3 ± 0.6 | 3.8 ± 0.6 | <0.001*** | 4.3 ± 0.6 | 3.9 ± 0.6 | <0.001*** |
| Ejection fraction, % | 60.7 ± 9.7 | 65.1 ± 7.3 | <0.001*** | 62.0 ± 8.6 | 64.5 ± 7.9 | <0.001*** |
| Hemoglobin, g/L | 145.4 ± 16.3 | 138.8 ± 16.5 | <0.0001*** | 144.8 ± 16.1 | 141.3 ± 16.1 | 0.006** |
| Uric acid, μmol/L | 362.9 ± 92.0 | 339.7 ± 88.9 | <0.0001*** | 358.0 ± 90.5 | 352.7 ± 92.2 | 0.462 |
| Creatinine, μmol/L | 83.8 ± 47.1 | 81.9 ± 33.9 | 0.484 | 84.1 ± 50.0 | 85.6 ± 41.1 | 0.673 |
| Cholesterol, mmol/L | 4.1 ± 1.0 | 4.1 ± 1.0 | 0.890 | 4.1 ± 1.0 | 4.1 ± 1.0 | 0.552 |
| Albumin, g/L | 41.3 ± 3.8 | 41.6 ± 3.6 | 0.238 | 41.4 ± 3.8 | 41.9 ± 3.6 | 0.094 |
| Homocysteine, μmol/L | 12.4 ± 5.6 | 11.6 ± 5.1 | 0.033* | 12.2 ± 5.3 | 11.7 ± 5.2 | 0.334 |
| C-rective protein, mg/L | 2.3 ± 4.5 | 2.9 ± 10.1 | 0.366 | 2.3 ± 4.5 | 3.1 ± 12.1 | 0.291 |
| RFCA related information and perioperative medication | ||||||
| Persistent AF | 298 (83.5) | 46 (8.5) | <0.001*** | 259 (82.7) | 29 (9.3) | <0.001*** |
| Com-CPVI | 190 (53.2) | 108 (20.0) | <0.001*** | 167 (53.4) | 73 (23.3) | <0.001*** |
| Amiodarone | 316 (88.5) | 481 (88.9) | 0.855 | 276 (88.2) | 275 (87.9) | 0.902 |
| Dronedarone | 22 (6.2) | 27 (5.0) | 0.449 | 19 (6.1) | 17 (5.4) | 0.731 |
| Propafenone | 11 (3.1) | 18 (3.3) | 0.838 | 10 (3.2) | 11 (3.5) | 0.824 |
| Beta blockers | 126 (35.3) | 143 (26.4) | 0.005** | 100 (31.9) | 93 (29.7) | 0.545 |
| Sacubitril valsartan | 63 (17.6) | 38 (7.0) | <0.001*** | 42 (13.4) | 29 (9.3) | 0.101 |
| SGLT2I | 7 (2.0) | 6 (1.1) | 0.296 | 5 (1.6) | 2 (0.6) | 0.447 |
| ACEI/ARB | 73 (20.4) | 132 (24.4) | 0.167 | 65 (20.8) | 79 (25.2) | 0.184 |
| Statins | 119 (33.3) | 196 (36.2) | 0.373 | 96 (30.7) | 122 (39.0) | 0.029* |
| Endpoints | ||||||
| Im-Recurr | 65 (18.2) | 63 (11.6) | 0.006** | 55 (17.6) | 34 (10.9) | 0.016* |
| Cost, 103 CNY | 78.2 ± 10.9 | 76.8 ± 6.7 | 0.020* | 77.7 ± 7.6 | 76.7 ± 6.7 | 0.066 |
| Duration of hospitalization, days | 5.3 ± 2.3 | 5.1 ± 2.0 | 0.062 | 5.2 ± 2.1 | 5.0 ± 1.8 | 0.092 |
| Duration of hospitalization after ablation, days | 3.1 ± 1.7 | 2.8 ± 1.2 | 0.001** | 3.1 ± 1.7 | 2.8 ± 1.2 | 0.005** |
LAD: Left atrial anteroposterior diameter; SGLT2I: Sodium-dependent glucose transporter 2 inhibitor; ACEI: Angiotensin converting enzyme inhibitor; ARB: Angiotensin receptor blocker. Power of Im-Recurr: 78%.
*P < 0.05, **P < 0.01 and ***P < 0.001.
The incidence of Im-Recurr in the IEC group was significantly higher than that in the control group (18.2% vs. 11.6%, P = 0.006). Even after PSM, the results still suggested that IEC was an independent predictor of Im-Recurr (17.6% vs. 10.9%, P = 0.016). Additionally, the hospitalization duration after ablation between groups also showed consistent statistical differences before and after PSM (Table 4).
When patients were grouped according to whether they received com-CPVI or CPVI, PSM analysis indicated no significant correlation of com-CPVI with Im-Recurr, cost, hospitalization duration, and hospitalization duration after ablation (Supplementary Table 6). Older age (≥ 65 years) was not associated with Im-Recurr, but older patients may experience prolonged hospitalization duration after ablation (Supplementary Table 7).
Follow Up
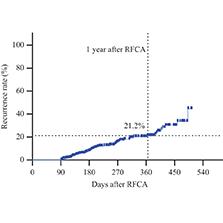
Follow-up information was collected from the electronic medical record system and analyzed. On the basis of our definition of loss to follow-up, 329 patients (36.6%) were excluded because their follow-up duration after RFCA was less than 90 days. The remaining 569 patients were followed up for a median duration of 187.0 ± 4.8 days. The follow-up results indicated a 1-year cumulative AF recurrence rate of 21.2% among patients with AF who underwent RFCA (Figure 1). Furthermore, the 1-year cumulative AF recurrence rate among patients with Im-Recurr after RFCA was significantly higher than that in the control group (47.0% vs. 15.8%, P < 0.0001) (Figure 2).
Discussion
The results of this study demonstrated that Im-Recurr after RFCA was significantly associated with greater costs and hospitalization durations among patients with AF. The 1-year cumulative AF recurrence rate in patients with Im-Recurr was significantly elevated, and IEC was an independent predictor of Im-Recurr.
Although RFCA is currently the most effective treatment for AF, the recurrence rate of AF remains high, thus affecting patient prognosis. Traditionally, the blanking period has been considered a transitional stage after RFCA, during which AF recurrence is common but is not considered true recurrence [17]. This perspective stems from the belief that blanking period AF recurrence might be caused by reversible processes such as inflammation and transient autonomic dysfunction [18, 19]. However, our analysis indicated that Im-Recurr was closely associated with greater costs and hospitalization durations.
Current large-scale clinical studies exploring different ablation techniques for AF, including pulsed field ablation, cryoballoon ablation, and RFCA have often defaulted to a 3-month blanking period for postoperative recovery without clinical significance [20–22]. However, increasing evidence suggests a significant correlation between blanking period AF recurrence and subsequent true recurrence [17, 23]. Some studies have also indicated that later blanking period AF recurrence is associated with greater likelihood of subsequent true recurrence [17, 24–26]. The most recent meta-analysis has even recommended considering a more appropriate range of 4 weeks as the blanking period [27]. Thus, clinical observations are advancing understanding of the blanking period after RFCA.
Recently, the American Heart Association issued a scientific statement proposing the concept of “acute AF,” which refers to AF occurring in the context of other disease episodes [28]. AF after cardiac surgery is a unique type of acute AF. Im-Recurr, defined herein as AF recurrence within 48 hours after RFCA, shares similarities with acute AF. In agreement with the American Heart Association scientific statement, we have observed that Im-Recurr has adverse medical economic effects and influences long-term prognosis. In our follow-up analysis, the 1-year cumulative recurrence rate of AF in patients with Im-Recurr was approximately three times higher than that in the control group. Im-Recurr, as a special case of blanking period AF recurrence after RFCA and of acute AF, warrants greater attention.
In both regression and PSM analyses, IEC was an independent predictor of Im-Recurr. Guidelines have suggested that RFCA for non-paroxysmal AF does not require intraoperative sinus rhythm recovery as the endpoint to avoid excessive ablation [3]. However, IEC is commonly used to restore sinus rhythm in patients who reach the endpoint of RFCA yet still exhibit AF rhythm. Our results indicated that compared to patients who do not require IEC during RFCA, patients receiving IEC were more likely to experience Im-Recurr in combination with a higher recurrence rate of AF. Therefore, restoring sinus rhythm as the endpoint of RFCA may be necessary. The issue of whether com-CPVI decreases the recurrence rate of AF remains controversial. Multiple studies have shown that linear ablation does not increase the success rate of RFCA [22, 29–31]. Magnetic resonance imaging guided substrate modification has also not achieved ideal results [32]. However, Marshall ethanol infusion ablation may decrease AF recurrence [13]. Further in-depth research on future AF ablation strategies is needed to extend current evidence. Restoring sinus rhythm as the endpoint of RFCA to the greatest extent possible, while avoiding excessive ablation, may effectively decrease the recurrence rate of AF.
In conclusion, this study analyzed the Im-Recurr phenomenon after RFCA in patients with AF, and elaborated on its medical economic effects, correlation with long-term AF recurrence, and predictors. Few studies have examined Im-Recurr. Limited evidence suggests that landiolol, amiodarone, and/or ibutilide may decrease the incidence of Im-Recurr [33, 34]. However, whether such treatments improve long-term prognosis remains unclear. Future research should explore the optimization of ablation strategies to decrease Im-Recurr and investigate whether postoperative drug inhibition of Im-Recurr might improve long-term prognosis. Obtaining relevant results will deepen understanding of Im-Recurr.
Conclusions
Im-Recurr significantly increases costs and hospitalization durations among patients with AF undergoing RFCA. IEC serves an independent predictor of Im-Recurr. The occurrence of Im-Recurr is associated with an elevated 1-year cumulative recurrence rate of AF.
Limitations
The perioperative medication information available in this study is relatively limited; moreover, data on dosages, and accurate start times and durations, are lacking. This limitation was inherent to the retrospective study design. Although the primary endpoint, Im-Recurr, might not be substantially affected by postoperative medication, related effects on long-term follow-up outcomes still must be considered.
The sample size in the PSM analysis for IEC lacked sufficient power. Therefore, in the future, larger sample sizes and more optimized research designs are needed to further confirm the findings of this study.
In the follow-up analysis, 36.6% of patients were excluded because they had not reached at least 90 days’ follow-up. In addition, this study lacked follow-up data on AF recurrence during the blanking period after discharge. Prospective cohorts are needed in the future to further validate the conclusions of this study.